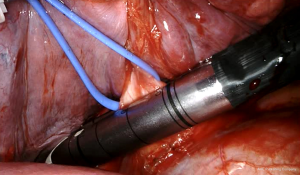
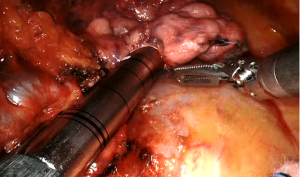
Robotic pneumonectomy
Introduction
Less invasive techniques, both robotic-assisted surgery and video-thoracoscopic thoracic surgery (VATS), gained popularity a worldwide diffusion in the thoracic domain largely used to perform lobectomy and segmentectomy in case of initial lung cancer (stage I and II) (1-5). More recently, robotic technique has also been adopted for advanced stages with good results in terms of morbidity, mortality and postoperative hospital stay (6-11).
Many reports on thoracic robotic approach described the technique, the advantages, and reported mainly data and solid experience in performing lobectomy (1-3,9-11) and segmentectomy (12-14) in early-stage lung cancer but very few centers performed robotic pneumonectomy (15-21).
Here, we sought to describe in this article our experience in performing robotic pneumonectomy, reporting indications, surgical technique, and pros and cons of this less invasive approach.
Indications
It is possible to perform a robotic pneumonectomy in case of tumors that extend through the fissures involving different lobes, in case of presence of adenopathy infiltrating the pulmonary artery and for which it is impossible to perform avascular resection/reconstruction, and in case of multiple primary tumors involving different lobes.
Preoperative evaluation
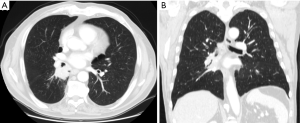
Computer tomography of the chest (Figure 1), abdomen and brain, and positron emission tomography is performed to complete the preoperative staging. Cardio-respiratory functional evaluation includes the following preoperative exams: spirometry, quantitative perfusion scan, electrocardiogram and echocardiogram. All patients receive diagnostic bronchoscopy while the assessment of mediastinal lymph nodes is performed by endobronchial ultrasound.
Surgical approach
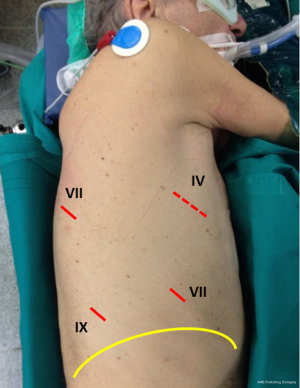
All patients are placed in lateral decubitus under general anesthesia with double lumen intubation. Patient’s position is reported in Figure 2; the description of the position has been previously reported (19). Briefly, the arm is positioned in front of the patient and the hip positioned at the same level of the chest allowing the camera to move without catching on the hip. The robot cart is always positioned at the patient’s head side (Figure 3), while the console is located in the same room. We used a complete four-arm approach is used to perform robotic pneumonectomy. As showed in Figure 2, a small mini-thoracotomy is performed anteriorly, in the IV intercostal space, with no costal spread. The camera port is positioned in the 7th intercostal space, and two other robotic ports are placed in the 8th-9th intercostal space, and at the 7th intercostal space in the posterior aspect of the scapula’s tip, respectively (Figure 2). The robot is docked, and the instruments introduced into the chest cavity under vision.
Vascular and bronchial isolation and resection
The first step for both the sides is the isolation of the inferior vein by a vessel loop performed after the resection of the inferior pulmonary ligament. The next step is the dissection of subcarinal lymph-nodes performed after the opening of the posterior mediastinal pleura. The inferior pulmonary vein is then sutured and sectioned by an EndoGia Roticulator with vascular cartridge introduced into the chest cavity through the utility incision. Since the use of the Da Vinci Xi, vascular and bronchial sutures are performed by an EndoWrist robotic stapler controlled at the console directly by the surgeon (Figure 4). Subsequently, the superior vein is isolated, encircled by a vessel loop, and sutured/resected by an endostapler or Endowrist stapler (Figure 5) inserted into the chest cavity through the postero-inferior trocar.
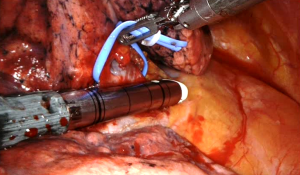
After the venous resection, it is easily visualizing the main pulmonary artery in both the sides. On the left side, the visualization and isolation of the pulmonary artery is made after the station #5 nodal dissection. Once the artery has been encircled by a vessel loop, the stapling articulated device (EndoGia or EndoWrist) is introduced by the postero-inferior port and the artery is transected (Figure 6).
The main bronchus is then isolated after the lymph node dissection of station #7; it is encircled by a vessel loop and transected by an endostapler or Endowrist stapler 45 mm green cartridge (Figure 7).
The lung is then removed by using an EndoCatch (Autosuture, Covidien) introduced in the chest cavity through the anterior mini-thoracotomy.
A short video reporting a left robotic pneumonectomy has been reported (22). This intervention was performed in a patient with a non-small cell lung cancer infiltrating the fissure. In this video the different steps of the surgical robotic procedures are reported (22).
On the right side, the para-tracheal nodal dissection (stations #2 and #4) is then performed. Sometimes a Harmonic scalpel may be used in patients with large amount of mediastinal fat for reducing bleeding and lymphatic leakage. Finally, one 32 Fr pleural drain is positioned through the camera port connected to a water seal device without suction for balancing the mediastinum.
Patients are usually awakened in the operative room. The chest drain is removed at the 3rd or 4th postoperative day if the mediastinum is balanced, and there is no bleeding.
Comment
A review of the English literature has been performed and a total of eleven robotic pneumonectomies have been attempted and only eight were completed by using robotic assistance (15-21) (Table 1). All the reported cases are case reports. The largest series is that one published by Galetta reporting 4 cases: 3 left and 1 right sided (19). Giulianotti (17) reported two conversions: one for “oncologic reasons”, and the other for a right sided pulmonary artery. Vidanapathirana reported the case of a female patient who was set up as a robotic sleeve resection but due to the intraoperative evidence of an extended tumor it was converted to a robotic pneumonectomy. This intervention was performed by a subxiphoid approach (20).

Full table
From a technical point of view, we think that the lateral position allows a better exposure of the pulmonary veins and artery during dissection and resection. We use an utility thoracotomy and do not use CO2. In our opinion, the utility thoracotomy is useful for different reasons: (I) it allows the assistant surgeon to directly aspirate fluids like blood and steam produced during coagulation; (II) it allows to bring in and take out the pleural cavity the removed lymph nodes and the gauzes used during the intervention, and, finally, (III) in case of a vascular injury and bleeding, it offers the possibility to control directly the surgical field from outside by the assistant surgeon.
In the first case of robotic pneumonectomy that we performed, we transected as first step the pulmonary veins and this determined a whole congestion of the lung rendering the procedure difficult due to the difficulties to move the lung. In the other 3 cases, we resected as first step the superior pulmonary vein, then the main pulmonary artery, and finally the inferior pulmonary vein avoiding the lung congestion and allowing to complete easily the procedure without technical inconveniences and with a reduction of the operative time.
As previously reported (19), in our experience of 4 cases of robotic pneumonectomy, the operative time ranged from 170 to 320 minutes (mean, 225 minutes). In all cases, patients were extubated in the operative room immediately after the operation; no patient spent time in intensive care unit. The postoperative stay ranged from 7 to 8 days, (mean, 7 days) with a number of resected lymph nodes ranging from 19 to 34 (mean, 25). Neither intraoperative nor postoperative complications occurred in our robotic pneumonectomy experience. To date, one patient is died, and 3 are alive, one with disease. The mean survival for the 4 patients is 86 months (range, 45–120 months).
With the advent of the robotic staplers (EndoWrist) and the new robotic instruments (Hem-o-lok and Harmonic scalpel) directly controlled by the surgeon at the console the robotic pulmonary resections have become simpler and the time of the surgical operation reduced.
From a technical point of view, in order to obtain a short bronchial stump, we suggest above all for the left pneumonectomy, to use a roticulator stapler with 45 mm cartridge which is get as far down as we could under the aortic arch.
Despite the small dimension of the utility thoracotomy, in our experience we had no problem in extracting the entire lung from the chest cavity by using Endobag without rib spreader, but performing a rocking and rotating motion of the bag. The difficulties in the extraction may be often due to the engorgement of the lung; to avoid this problem, we suggest to leave the inferior veins intact until the main artery is divided.
Another important point to underline is the completeness of resection which is obtained in our experience in all cases; in fact, it is equivalent to that of open surgery, including the completeness of lymph node dissection of the different nodal stations both on the right and left side.
Robotic pneumonectomy is not frequently performed probably due to complexity of the procedure and the experience of the operators. The recent ability demonstrated by some authors to perform robotic lung-sparing surgery (23-26) has opened new frontiers also in the robotic domain. These new surgical less invasive approaches should be reserved for centers with high volume of robotic surgical operations.
Acknowledgments
Funding: This work was partially supported by the Italian Ministry of Health with Ricerca Corrente and 5x1000 funds.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Lorenzo Spaggiari, Luca Bertolaccini) for the series “The Role of Pneumonectomy in Thoracic Surgery in The Third Millennium” published in Shanghai Chest. The article was sent for external peer review organized by the Guest Editor and the editorial office.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/shc.2020.04.01). The series “The Role of Pneumonectomy in Thoracic Surgery in The Third Millennium” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Park BJ, Flores RM, Rush VW, et al. Robotic assistance for video-assisted thoracic surgical lobectomy: technique and initial results. J Thorac Cardiovasc Surg 2006;131:54-9. [Crossref] [PubMed]
- Veronesi G, Galetta D, Maisonneuve P, et al. Four-arm robotic lobectomy for the treatment of early stage lung cancer. J Thorac Cardiovasc Surg 2010;140:19-25. [Crossref] [PubMed]
- Casiraghi M, Galetta D, Borri A, et al. Ten Years' Experience in Robotic-Assisted Thoracic Surgery for Early Stage Lung Cancer. Thorac Cardiovasc Surg. 2019;67:564-72. [Crossref] [PubMed]
- Gopadas RR, Bakaeen FG, Dao TK, et al. Video-assisted thoracoscopic versus open thoracotomy lobectomy in a cohort of 13,619 patients. Ann Thorac Surg 2010;89:1563-70. [Crossref] [PubMed]
- Whitson BA, Groth SS, Duval SJ, et al. Surgery for early-stage non-small cell lung cancer: a systemic review of the video-assisted thoracoscopic surgery versus thoracotomy approached to lobectomy. Ann Thorac Surg 2008;86:2016-8. [Crossref]
- Cao C, Manganas C, Ang SC, et al. A systematic review and meta-analysis on pulmonary resections by robotic video-assisted thoracic surgery. Ann Cardiothorac Surg 2012;1:3-10. [PubMed]
- Kent M, Wang T, Whyte R, et al. Open, video-assisted thoracic surgery, and robotic lobectomy: review of a national database. Ann Thorac Surg 2014;97:236-42; discussion 242-4. [Crossref] [PubMed]
- Oh DS, Reddy RM, Gorrepati ML, et al. Robotic-assisted, video-assisted thoracoscopic and open lobectomy: propensity-matched analysis of recent premier data. Ann Thorac Surg 2017;104:1733-40. [Crossref] [PubMed]
- Mariolo AV, Casiraghi M, Galetta D, et al. Robotic Hybrid Approach for an Anterior Pancoast Tumor in a Severely Obese Patient. Ann Thorac Surg 2018;106:e115-e116. [Crossref] [PubMed]
- Casiraghi M, Sedda G, Diotti C, et al. Postoperative outcomes of robotic-assisted lobectomy in obese patients with non-small-cell lung cancer. Interact Cardiovasc Thorac Surg. 2020;30:359-65. [Crossref] [PubMed]
- Spaggiari L, Sedda G, Maisonneuve P, et al. A Brief Report on Survival After Robotic Lobectomy for Early-Stage Lung Cancer. J Thorac Oncol. 2019;14:2176-80. [Crossref] [PubMed]
- Cerfolio RJ, Watson C, Minnich DJ. One hundred planned robotic segmentectomies: early results, technical details, and preferred port placement. Ann Thorac Surg. 2016;101:1089-95. [Crossref] [PubMed]
- Wei B, Cerfolio R. Technique of robotic segmentectomy. J Vis Surg 2017;3:140. [Crossref] [PubMed]
- Nguyen D, Gharagozloo F, Tempesta B, et al. Long-term results of robotic anatomical segmentectomy for early-stage l non-small-cell lung cancer. Eur J Cardiothorac Surg. 2019;55:427-33. [Crossref] [PubMed]
- Spaggiari L, Galetta D. Pneumonectomy for lung cancer: a further step in minimally invasive surgery. Ann Thorac Surg 2011;91:e45-7. [Crossref] [PubMed]
- Rodriguez JR. Total port robotic pneumonectomy. Gen Thorac Cardiovasc Surg 2013;61:538-41. [Crossref] [PubMed]
- Giulianotti PC, Buchs NC, Caravagios G, et al. Robot-Assisted lung resection: outcomes and technical details. Interact Cardiovasc Thorac Surg 2010;11:388-92. [Crossref] [PubMed]
- Khan N, Fikfak V, Chan E, et al. “Five on a dice” port placement allows for successful robot-assisted left pneumonectomy. Thorac Cardiovasc Surg Rep 2017;6:e42-e44. [Crossref] [PubMed]
- Galetta D, Casiraghi M, Spaggiari L. Advanced resections in minimally invasive surgery: robotic pneumonectomy. Shanghai Chest 2019;3:25. [Crossref]
- Vidanapathirana CP, Papoulidis P, Nardini M, et al. Subxiphoid robotic-assisted right pneumonectomy. J Thorac Dis 2019;11:1629-31. [Crossref] [PubMed]
- Dylewski MR, Ohaeto AC, Pereira JF. Pulmonary resection using a total endoscopic robotic video-assisted approach. Semin Thorac Cardiovasc Surg 2011;23:36-42. [Crossref] [PubMed]
- Galetta D, Casiraghi M, Spaggiari L. A case of left robotic pneumonectomy performed for an interlobar non-small cell lung cancer infiltrating both the left pulmonary lobes. Asvide 2019;6:119. Available online: http://www.asvide. com/article/view/31234.
- Qiu T, Zhao Y, Xuan Y, et al. Robotic-assisted double-sleeve lobectomy. J Thorac Dis 2017;9:E21-E25. [Crossref] [PubMed]
- Li C, Zhou B, Han Y, et al. Robotic sleeve resection for pulmonary disease. World J Surg Oncol 2018;16:74. [Crossref] [PubMed]
- Shanahan B, O'Sullivan KE, Redmond KC. Robotic sleeve lobectomy-recent advances. J Thorac Dis 2019;11:1074-5. [Crossref] [PubMed]
- Galetta D, Casiraghi M, Pardolesi A, et al. New stapling devices in robotic surgery. J Vis Surg 2017;3:45. [Crossref] [PubMed]
Cite this article as: Galetta D, Spaggiari L. Robotic pneumonectomy. Shanghai Chest 2021;5:6.